Introduction: water electrolysis method without membrane
Researchers in Sweden recently published research on electrolyzer for membrane-free water electrolysis and presented groundbreaking findings on sustainable hydrogen production that can replace fossil fuels. This innovative SuperCapacitive Electrolyzer (SCE) enables membrane-free operation and addresses the critical challenges of scalability and cost reduction of green hydrogen production using earth-abundant materials.
The key to SCE’s breakthrough is the spatial and temporal separation of hydrogen and oxygen production, improving safety and efficiency over traditional water electrolysis methods. Using a dual-functional catalyst and cell optimization, the researchers achieved an impressive energy efficiency of 69% at real current densities.
*U.S. Department of Energy (DoE), PEM Water Electrolysis efficiency target: 69% (LHV)
SCE operates as two identical cells, enabling continuous hydrogen production. This system allows the production of hydrogen and oxygen to be separated into separate steps utilizing dual-function electrodes for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in separate chambers, and this separation is achieved through a supercapacitor storage mechanism. Eliminates the need for membranes facilitated by charging and discharging of auxiliary electrodes, mimicking .
And to further validate the SCR water electrolysis system, an electrocatalyst with high activity for both HER and OER is needed. Among the various catalysts studied for electrocatalysis, transition metal-based phosphides, especially cobalt iron phosphide (CoFeP), boast high activity and stability. Dual-functional catalysts that can accelerate both hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) are critical to the efficiency of water electrolysis systems.
CoFeP stands out due to its high activity and stability in alkaline conditions, synthesized via galvanostatic electrodeposition method on Ni foam. This process creates a uniform, dense layer of CoFeP, which aids in the catalytic process. The electrochemical performance of CoFeP evaluated through linear sweep voltammetry (LSV) in alkaline media shows superior activity for both hydrogen evolution and oxygen evolution reactions compared to single metal phosphides. This performance is attributed to the modulation of the electronic structure around the Co active site by Fe atoms.
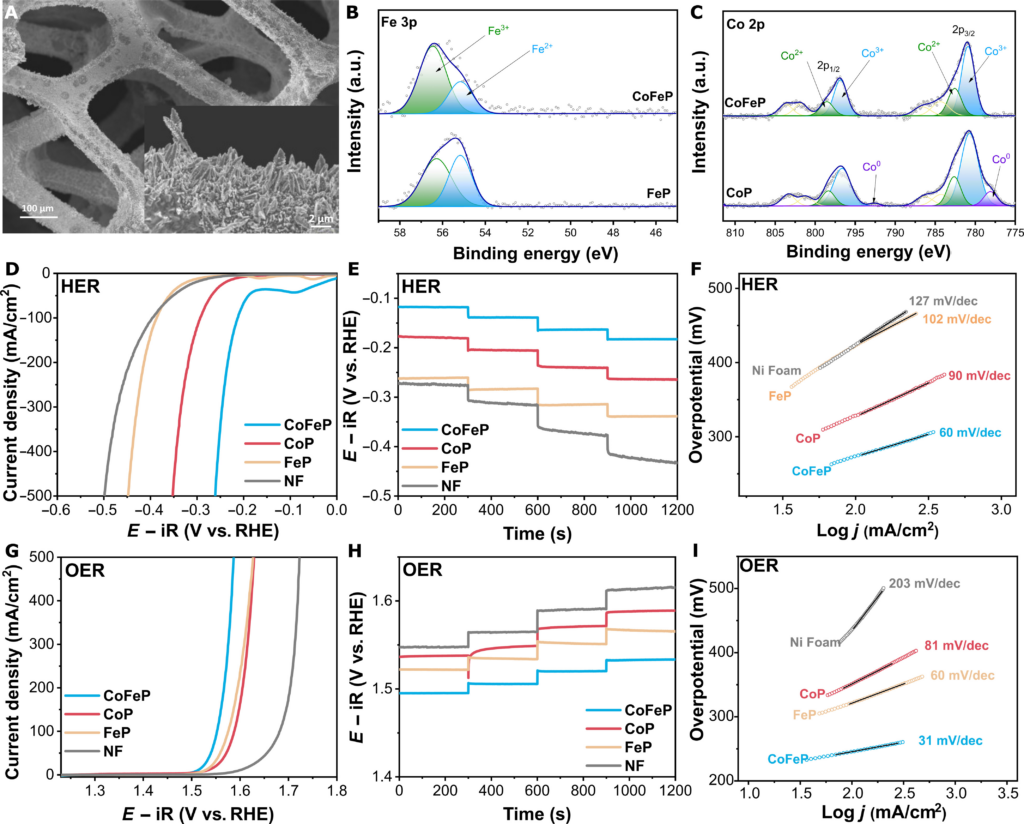
And through single cell evaluation, the SCE system achieved Faraday efficiencies of up to 99%, demonstrating efficient reaction conversion. The energy consumption obtained for full water electrolysis in experiments using a bifunctional catalyst (CoFeP) shows similar cell voltage and efficiency values to noble and transition metal-based catalysts reported in the literature.
This study demonstrated the potential of a membrane-free water electrolysis system utilizing abundant and low-cost materials. And its possible operation in both acidic and alkaline conditions confirms its adaptability and potential for a variety of applications.
Researchers are tirelessly working to enhance the commercial viability of SCE, exploring avenues to optimize its performance and efficiency. One key area of focus is increasing the capacitive storage capacity of the electrodes, which plays a crucial role in maximizing the energy density and overall output of the system. Additionally, efforts are underway to optimize electrode conductivity, aiming to reduce resistance and minimize energy losses during the hydrogen production process.
By addressing these key areas, researchers aim to further improve the energy efficiency of SCE, making it an increasingly attractive and cost-effective solution for green hydrogen production. Reduced operating costs and enhanced sustainability will not only benefit the energy sector but also contribute to the broader goal of transitioning towards a hydrogen-based economy.
Conclusion
The development of SCE represents a significant step forward in the quest towards sustainable and efficient hydrogen production. SCE’s development for green hydrogen production is one of the ways to combat carbon neutrality and climate change and accelerate the realization of a hydrogen society. Researchers will pursue research aimed at improving the commercial viability of SCE and will work on potential areas for improvement increasing the capacitive storage capacity of the electrodes and optimizing electrode conductivity to reduce resistance.
This is expected to further improve energy efficiency and reduce operating costs. The development of SCE represents a significant step forward in the quest towards sustainable and efficient hydrogen production. SCE’s development for green hydrogen production is one of the ways to combat carbon neutrality and climate change and accelerate the realization of a hydrogen society. Researchers will pursue research aimed at improving the commercial viability of SCE and will work on potential areas for improvement increasing the capacitive storage capacity of the electrodes and optimizing electrode conductivity to reduce resistance.
This is expected to further improve energy efficiency and reduce operating costs. The development of SCE represents a significant step forward in the quest towards sustainable and efficient hydrogen production. SCE’s development for green hydrogen production is one of the ways to combat carbon neutrality and climate change and accelerate the realization of a hydrogen society.
Researchers will pursue research aimed at improving the commercial viability of SCE and will work on potential areas for improvement increasing the capacitive storage capacity of the electrodes and optimizing electrode conductivity to reduce resistance. This is expected to further improve energy efficiency and reduce operating costs.
Source: Toledo-Carrillo, Esteban A., et al. “Decoupled supercapacitive electrolyzer for membrane-free water splitting.” Science Advances 10.10 (2024): eadi3180.
https://www.science.org/doi/10.1126/sciadv.adi3180
※ This report has been compiled for the purpose of providing general information. It is based on data gathered by CHEMiFORGE. Should you have any inquiries or need to make decisions based on this report, it is advisable to consult with a CHEMiFORGE.